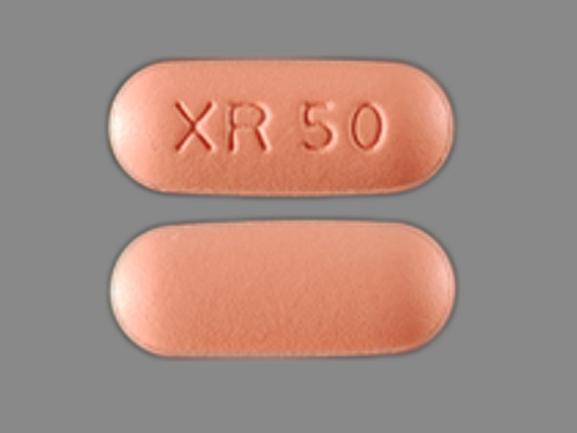
Summary Product NRA-Quetiapine XR Issue Health products – Product quality What to do Consult your healthcare provider prior to discontinuing use of the affected product, or for any health concerns. Distribution National Affected products Filter items Showing 1 to 1 of 1 entries Show 102550100 entries Brand Product Name Market Authorization Dosage Form Strength Lot numbers NORA PHARMA INC NRA-Quetiapine XR DIN …
Souvenir du Canada Plush Toys recalled
Summary Product Souvenir du Canada Plush Toys Issue Consumer products – Choking hazard What to do Immediately stop using the recalled products and dispose of them. Affected products This recall involves the Souvenir du Canada plush toys including the Brown Bear, White Bear, Moose and Black Bear. The plush toys have a red and white hat and sweater with Canada …
Believe Supplements products recall: Contamination (milk derived proteins)
Summary Product Believe Supplements products Issue Health products – Product quality What to do Consult your health care professional if you have any health concerns. Issue Affected lot(s) may have come into contact with milk derived proteins during manufacturing. What you should do Verify if your product is affected. Consult your healthcare provider prior to discontinuing use of the affected …
Sambucus Organic Cold & Flu syrup-Out of specification
Summary Product Sambucus Organic Cold & Flu syrup Issue Health products – Product quality What to do Consult your health care professional if you have any health concerns. Affected products Filter items Showing 1 to 1 of 1 entries Show 102550100 entries Brand Product Name Market Authorization Dosage Form Strength Lot numbers Nature’s Way of Canada Sambucus Organic Cold & Flu syrup …
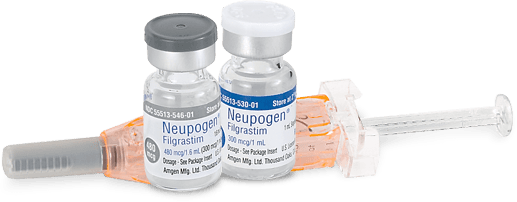
Neupogen vials: Out of specification (Stability)
Summary Product Neupogen 300 mcg/mL (480 mcg /1.6 mL) vials Issue Health products – Product quality What to do Consult your health care professional if you have any health concerns. Affected products Filter items Showing 1 to 4 of 4 entries Show 102550100 entries Brand Product Name Market Authorization Dosage Form Strength Lot numbers Amgen Canada Neupogen 300 mcg/mL (480 mcg /1.6 …
Lolanta Children’s Bathrobes recalled due to burn hazard
Summary Product Lolanta Children’s Bathrobes Issue Consumer products – Burn hazard What to do Immediately stop using the recalled product and contact Nanchang Zhongcangjishi E-commerce for a full refund. Affected products This recall involves Lolanta 100% polyester hooded dinosaur children’s bathrobes. The flannel bathrobes are green and have attached belts and patch pockets. The bathrobes were sold in sizes 2-3 …
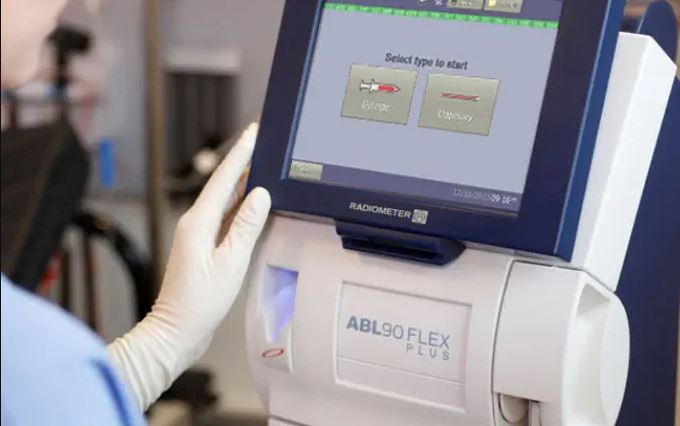
All you need to know about ABL90 Flex and Flex Plus
Product ABL90 Flex and Flex Plus Issue Medical devices – Performance What to do Contact the manufacturer if you require additional information. Affected products Filter items Showing 1 to 2 of 2 entries Show 102550100 entries Affected products Lot or serial number Model or catalog number ABL90 Flex RWIN10 version 1.1 (933-894) RWIN10 version 1.0 (933-803) 393-090 ABL90 Flex Plus RWIN10 version …
Unauthorized sexual enhancement products may pose serious health risks
Product Various unauthorized health products promoted for sexual enhancement Issue Health products – Unauthorized product What to do Stop using these products and consult your health care professional if you have health concerns. Report these or any unauthorized health products to Health Canada. Affected products Filter items Showing 1 to 10 of 432 entries Show 102550100 entries Photo Product Hazard Identified Company Action Taken …
Canada signs deal with Nova Scotia to improve access to drugs for rare diseases
Nova Scotia – In Canada, one in 12 people live with a rare disease, and for most people affected, the cost of treatment is unaffordable. Canadians deserve a health care system that provides timely access to quality health services and medications, including effective drugs for rare diseases, regardless of where they live or their ability to pay. Kamal Khera, Canada’s …
Travelling for March Break? CBSA gives tips for a smooth trip
Ottawa – The Canada Border Services Agency (CBSA) reminds travellers that it can be extra busy at the border during March Break. Every day, the CBSA works hard to protect Canadians, support the economy and ensure the safe and efficient movement of people and goods across the border. In 2024, we welcomed over 93.4 million travellers, stopped over 34,400 kg of illegal drugs …